4D Yoga Portugal Retreat: 22nd – 28th Sept 2024

Leonardo Da Vinci

Leonardo Da Vinci


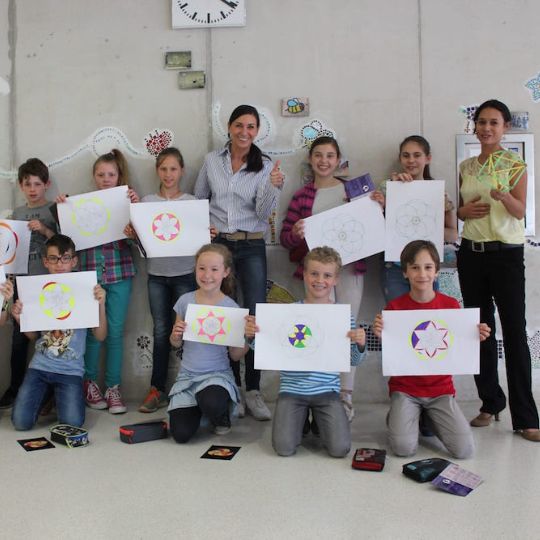

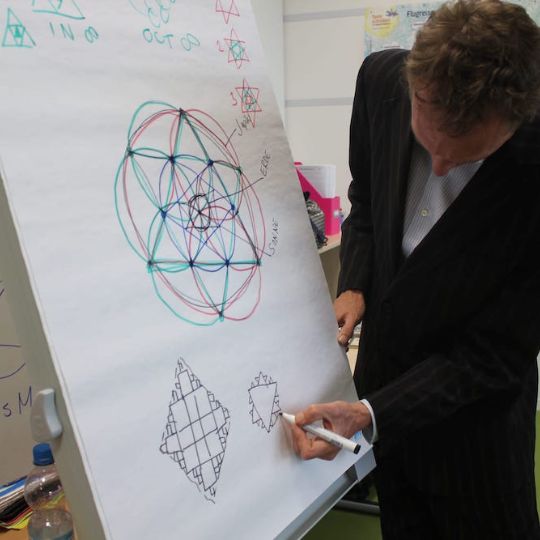

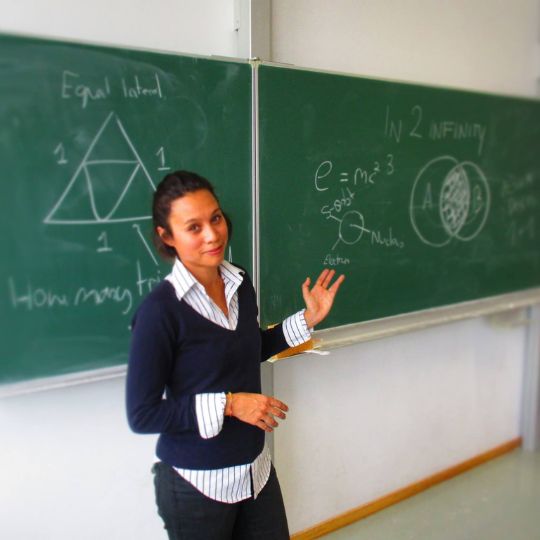



Geometry is the key to Education
Step into the footsteps of the greatest minds of human history, and just like genius Leonardo da Vinci, use a drawing compass and ruler to comprehend how reality works.
Space
Everything can be visualized in space, i.e. geometry
Art
Accelerated learning through geometric constructions
Model
Build a mental image to remember better
Da Vinci School Founders
New learning methods
We have developed tactile learning methods based on our new discoveries currently being published on in2infinity that will accelerate your understanding of the world.
The Da Vinci school integrates the 7 Liberal Arts of the Ancient Greeks to approach various subjects such as chemistry, physics, biology, maths, music and philosophy in an interdisciplinary and creative way. All knowledge is expressed through drawing compass construction.
The Da Vinci School incorporates Accelerated Learning techniques, which are based on the way we naturally use our brain. By involving the whole body with tools such as art, music, and relaxation, it increases our capacity of learning.
The Da Vinci School offers online courses and offline events. Join our community of teachers and students around the world and learn in your own time and space.
Learn from a teacher of your choice who will guide you through the content of our curriculum and experience the live environment with other students.
Free Online courses
Start your geometric journey here with our selection of free courses.

Inspiring Events
Whether in person or online, our team is bringing conscious connection and inspiring ideas to beautiful locations worldwide. Currently, we are in Tulum, Mexico. If you’re in town, come and see us!

Retreats & Trainings
Embark on a transformative journey where ancient wisdom seamlessly intertwines with cutting-edge science, all rooted in the mesmerizing tapestry of the Geometric Universe.

Workshops & Courses
Dive into hands-on learning with our creative Sacred Geometry workshops and courses! Unlock the mysteries of the universe in a fun and accessible way suitable for all ages.

Talks & Webinars
Explore the intricacies of our groundbreaking discoveries and join the discussion as we are solving modern scientific enigmas to unveil deep philosophical insights.
Transformative Journeys
4D Yoga Retreats
Next : Bacalar, Mexico (27th Feb 24)
Gain access to the matrix and change your perception of reality in our immersive retreats, where spiritual practises meet science through art.
Book out now!
The Geometric Universe – Beyond space and time
This book unveils the Geometric Theory of the cosmos, from the atom to the galaxy. Filled with intricate illustrations, it offers a concise exploration of the Multidimensional Universe, challenging our understanding of space, time, and existence.

Online Shop
Grab your geometric goodies!
Discover a wealth of inspiration at our online shop! From visually stunning educational posters to captivating geometric mind games and original art, immerse yourself in a collection designed to unravel the mysteries of the Geometric Universe.
what people say...

















